Quickly Implement Flow Cytometer Designs Using High-Accuracy Data Acquisition Modules
Contributed By DigiKey's North American Editors
2022-04-21
Flow cytometry is widely used by clinicians and diagnosticians to analyze cell characteristics. One cell at a time, they optically evaluate protein levels, blood health, granularity, and cell size, among other attributes. Though they are highly sensitive systems, designers of cytometers are under constant pressure to accelerate the analysis time, requiring new approaches to both flow cytometry and its associated electronics.
Cytometers subject individual cells to laser light to create scattered and fluorescence signals. To quickly and accurately capture the resulting light and convert it to digital signals requires an avalanche photodiode (APD) and complex electronics. The circuitry for this process can take a long time to design and implement, particularly given that flow cytometry data acquisition systems require high-speed, low-noise devices to ensure system accuracy.
To cost-effectively support faster flow cytometry analysis, designers can resolve speed and accuracy issues with a data acquisition solution comprising internal amplifier drivers and an analog-to-digital converter (ADC).
This article will briefly show how flow cytometry systems work. It then introduces Analog Devices’ ADAQ23878, an 18-bit ADC module, and shows how it can be used to design a flow cytometer detection and conversion stage. An associated evaluation kit will also be introduced.
Modern flow cytometry principles
Modern flow cytometry is an automated process that analyzes cell and surface molecules, characterizes, and defines different cell types in a heterogeneous cell population. Not counting the preparation time, which could be more than an hour, the instrument performs a three to six characteristic assessment of 10,000 single cells in less than a minute.
To enable this, flow cytometry's single-cell preparation step is critical. The organization of the samples occurs in a sheath fluid hydrodynamically to focus cells or particles into a narrow, single-cell line sample stream for analysis. With this transformation, the single cells must maintain their natural biological characteristics and biochemical components.
Figure 1 shows a diagram of a flow cytometer instrument that starts at the top with the multiple cell sample.
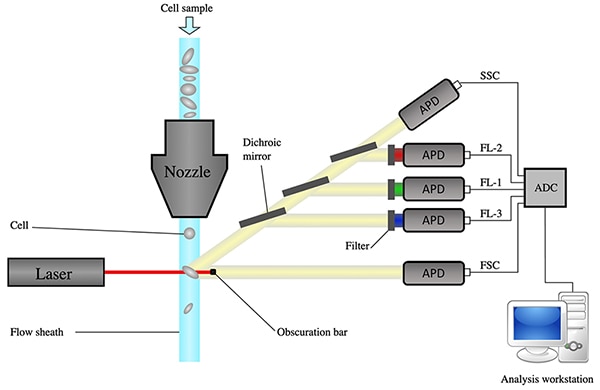 Figure 1: Diagram of a flow cytometer, from sheath focusing to data acquisition. (Image source: Wikipedia, modified by Bonnie Baker)
Figure 1: Diagram of a flow cytometer, from sheath focusing to data acquisition. (Image source: Wikipedia, modified by Bonnie Baker)
The six main components of the flow cytometer are a flow cell, a laser, an avalanche photodiode (APD), a transimpedance amplifier (TIA), an ADC, and a computer for data collection and analysis.
The flow cytometer has a liquid stream or sheath fluid, which is narrowed to carry and align the cells into a single file through the light beam. The laser light captures one cell at a time, creating a forward-scattered light (FSC) signal and a side-scattered light (SSC) signal. Fluorescence light is sorted using mirrors and filters and then amplified by an APD.
The next step is to detect, digitize, and analyze the resulting light output after it hits the APD. For detection, the Analog Devices LTC6268 500 megahertz (MHz) ultra-low bias current, low voltage noise FET input op amp is ideal for the high-speed TIA needed for detection.
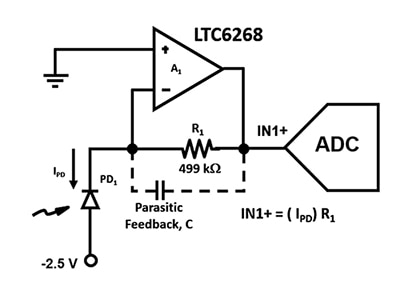 Figure 2: The TIA circuit uses an APD (PD1) and a low input current FET op amp to convert ultra-low photodiode currents to an output voltage at IN1+. (Image source: Bonnie Baker)
Figure 2: The TIA circuit uses an APD (PD1) and a low input current FET op amp to convert ultra-low photodiode currents to an output voltage at IN1+. (Image source: Bonnie Baker)
It is essential to design this amplifier circuit with the widest bandwidth possible, so parasitic capacitances must be minimized. For example, the parasitic feedback capacitance, C, influences Figure 2’s circuit stability and bandwidth. Regardless of the resistor package choice, there will always be a parasitic capacitance in the amplifier’s feedback path. However, an 0805 package, which has a longer distance between the end caps and the lowest parasitic capacitance, is preferable for high-speed applications.
Increasing the distance between R1’s end caps is not the only way to decrease capacitance. Another way to reduce plate-to-plate capacitance is to shield the E field paths that give rise to the parasitic capacitance by placing an extra ground trace underneath the resistance, R1 (Figure 3).
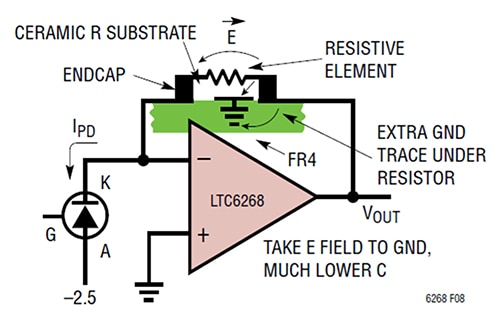 Figure 3: Adding a ground trace under the feedback resistor shunts the E field away from the feedback side and dumps it to ground. (Image source: Analog Devices)
Figure 3: Adding a ground trace under the feedback resistor shunts the E field away from the feedback side and dumps it to ground. (Image source: Analog Devices)
In this instance, the method specifically involves placing a short ground trace below and between the resistor pads near the TIA’s output end. This technique renders a parasitic capacitance value of 0.028 picofarads (pF) with a TIA bandwidth of 1/(2π*RF*CPARASITIC), equaling 11.4 MHz.
The optical light signals point toward several avalanche diodes with appropriate optical filters. The APD, TIA, and ADC system convert these signals to their digital representation and send the data to the microprocessor for further analysis.
Modern instruments usually have multiple lasers and APDs. The current commercial devices have ten lasers and thirty avalanche photodiodes. Increasing the laser and photomultiplier detector number allows multiple anti-body labeling to precisely identify target populations by phenotypic markers.
Still, the speed of analysis depends on a fine balance of:
- The fluid sheath velocity
- The ability of the hydrodynamic focus process to form single-cell lines
- The tunnel diameter
- The ability to preserve a cell’s integrity
- The electronics
Flow cytometry acoustic focusing
While adding multiple lasers and APDs accelerates analysis and identification, at best, the latest modern single-cell flow cytometry methods can collect data on up to one million individual cells per minute. In many applications, such as detecting circulating tumor cells present in blood at levels as low as 100 cells per milliliter, this is inadequate. In rare-cell clinical applications, tests regularly require the time-consuming analysis of billions of cells.
The alternative to the hydrodynamically focused cell preparation process is an acoustic focusing process. Here, a piezoelectric material, such as lead zirconate titanate (PZT), is attached to a glass capillary to convert electrical pulses into mechanical vibrations (Figure 4a). By using a PZT to vibrate the sidewalls of a glass capillary at the rectangular flow cell’s resonant frequency, the system generates a variety of acoustic standing waves with varying numbers of pressure nodes.
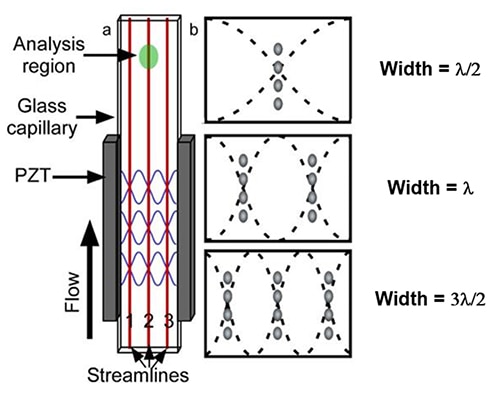 Figure 4: An illustration of an acoustic flow cell made with a rectangular glass capillary (a). The location of the first three pressure nodes for a fixed-width capillary (b). (Image source: National Center for Biotechnology Information)
Figure 4: An illustration of an acoustic flow cell made with a rectangular glass capillary (a). The location of the first three pressure nodes for a fixed-width capillary (b). (Image source: National Center for Biotechnology Information)
These PZT frequency nodes align flowing particles in multiple, discrete streamlines (Figure 4b). The acoustic flow cell uses a linear, standing acoustic wave to tune into various wavelengths by creating single or multiple harmonics. As predicted by the simple linear standing wave model, the cells in the sample produce single or numerous single-cell lines within the flow chamber.
With this precise organization of cells, the width of the flow sheath tunnel can widen to allow faster flow rates past the laser beam (Figure 5).
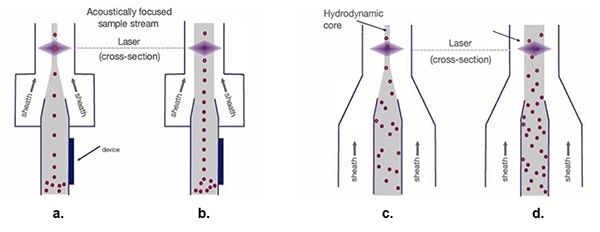 Figure 5: With the hydrodynamic sample stream (c. and d.), as the sheath width increases, the cell samples scatter, making the optical measurement process difficult. Acoustically focused sample streams (a. and b.) maintain single-file cells regardless of the sheath width. (Image source: Thermo Fischer Scientific)
Figure 5: With the hydrodynamic sample stream (c. and d.), as the sheath width increases, the cell samples scatter, making the optical measurement process difficult. Acoustically focused sample streams (a. and b.) maintain single-file cells regardless of the sheath width. (Image source: Thermo Fischer Scientific)
Traditional hydrodynamic focusing (Figure 5c.) arranges the single-cell lines in preparation for laser scanning. While a wider funnel for the sample stream core allows a higher sheath material velocity (Figure 5d.), it also results in the spread of the single-cell organization, producing signal variation and compromised data quality.
Acoustic focusing (Figure 5a.) positions biological cells and other particles in tight alignment, even with a wider tunnel. This precise cell alignment allows higher sample rates while maintaining data quality (Figure 5b.).
In practice, flow cytometry acoustic focusing increases cell sampling frequency by ~20x (Figure 6).
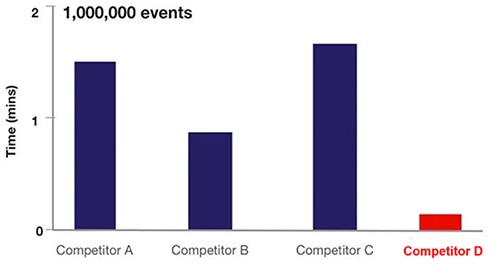 Figure 6: Comparison of sampling time for various flow cytometry equipment based on fluid flow cytometry (A, B, C) versus acoustic focusing cytometry (D). (Image source: Thermo Fischer Scientific)
Figure 6: Comparison of sampling time for various flow cytometry equipment based on fluid flow cytometry (A, B, C) versus acoustic focusing cytometry (D). (Image source: Thermo Fischer Scientific)
In Figure 6, equipment from A, B, and C uses hydrodynamic technology, while D uses the acoustic focusing cytometry flow approach.
Acoustic focusing flow cytometry data acquisition
The design of the electronics for acoustic focusing flow cytometry equipment requires high-speed photo-sensing electronics to accommodate the speed of the blood cells and sheath fluid through the larger diameter nozzle. The 600 MHz high-speed LTC6268 mentioned earlier, in combination with a specialized 0805 resistor package layout, brings the optical sensing rate up to 11.4 MHz (Figure 7, left). The output of the LTC6268 is fed to an Analog Devices ADAQ23878 ADC for digitization.
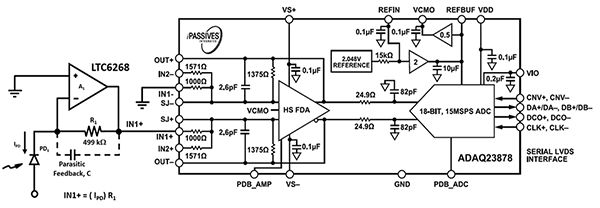 Figure 7: The ADAQ23878 ADC digitizes the optical signal from the photodiode (PD1) and the TIA circuit (left). (Image source: Bonnie Baker)
Figure 7: The ADAQ23878 ADC digitizes the optical signal from the photodiode (PD1) and the TIA circuit (left). (Image source: Bonnie Baker)
The ADAQ23878 is an 18-bit, 15 mega samples per second (MSPS), precision, high-speed system-in-package (SIP) data acquisition solution. It greatly reduces the development cycle of precision measurement systems by transferring the design burden of input driver component selection, optimization, and layout from the designer to the device.
The SIP’s modular approach reduces end system component count by combining multiple common signal processing and conditioning blocks in a single device, along with the high-speed, 18-bit, 15 MSPS successive approximation register (SAR) ADC. These blocks include a low-noise, fully-differential ADC driver amplifier, and a stable reference buffer.
The ADAQ23878 also incorporates the critical passive components that use Analog Devices’ iPassive technology to minimize temperature-dependent error sources and optimize performance. The ADC’s fast-settling driver stage contributes to its ability to ensure rapid data acquisition.
Evaluating the ADAQ23878 µModule
To evaluate the ADAQ23878, Analog Devices provides the EVAL-ADAQ23878FMCZ evaluation board (Figure 8). The board demonstrates the performance of the ADAQ23878 μModule and is a versatile tool for evaluating a flow cytometry front-end design, and a variety of other applications.
 Figure 8: The EVAL-ADAQ23878FMCZ evaluation board for the ADAQ23878 has power circuits on board, comes with associated software for control and data analysis, and is SDP-H1 compatible. (Image source: Analog Devices)
Figure 8: The EVAL-ADAQ23878FMCZ evaluation board for the ADAQ23878 has power circuits on board, comes with associated software for control and data analysis, and is SDP-H1 compatible. (Image source: Analog Devices)
The EVAL-ADAQ23878FMCZ evaluation board requires a personal computer running Windows 10 or higher, a low noise, precision signal source, and a band-pass filter suitable for 18-bit testing. The evaluation board needs the ADAQ23878 ACE plugin and SPD-H1 driver.
Conclusion
The examination of one biological cell at a time using standard hydrodynamic focus flow cytometry techniques has been successful, but with the need for faster analysis, there has been a shift to techniques based on acoustic focus flow methods. However, the electronics supporting more advanced flow cytometry must also improve, while minimizing space, cost, and development time.
As shown, the LTC6268 high-speed op amp and the ADAQ233878 precision, high-speed, μModule data acquisition solution can be combined to create the complete data acquisition system for advanced flow cytometry equipment.

Disclaimer: The opinions, beliefs, and viewpoints expressed by the various authors and/or forum participants on this website do not necessarily reflect the opinions, beliefs, and viewpoints of DigiKey or official policies of DigiKey.




